Research in the Schröder lab focuses on host-microbe interaction in the intestine and can be summarised by the acronym MUCUBACTER: MUCosa, gut BACTERia, MUCUs and antiBACTERial peptides.
We use a combination of gut microbiota analyses, microbiota transplantations (FMT), state-of-the-art ex vivo mucus measurements, antimicrobial assays, as well as confocal and fluorescence microscopy. In addition, we use dietary interventions in mice and humans and closely work with Norrlands University Hospital Umeå (NUS) to analyze relevant patient samples.
The intestine is one of the most densely populated habitats on our planet. Trillions of microorganisms, mainly bacteria, live in our gut in a relationship from which both sides benefit: The host provides continuous supply of nutrients and a constantly warm temperature, while the bacteria produce vitamins and other factors that are beneficial for the host. However, the high density of the gut bacteria is a permanent threat to the body and efficient defence mechanisms are required to protect the body against infection by this intestinal bacterial community.
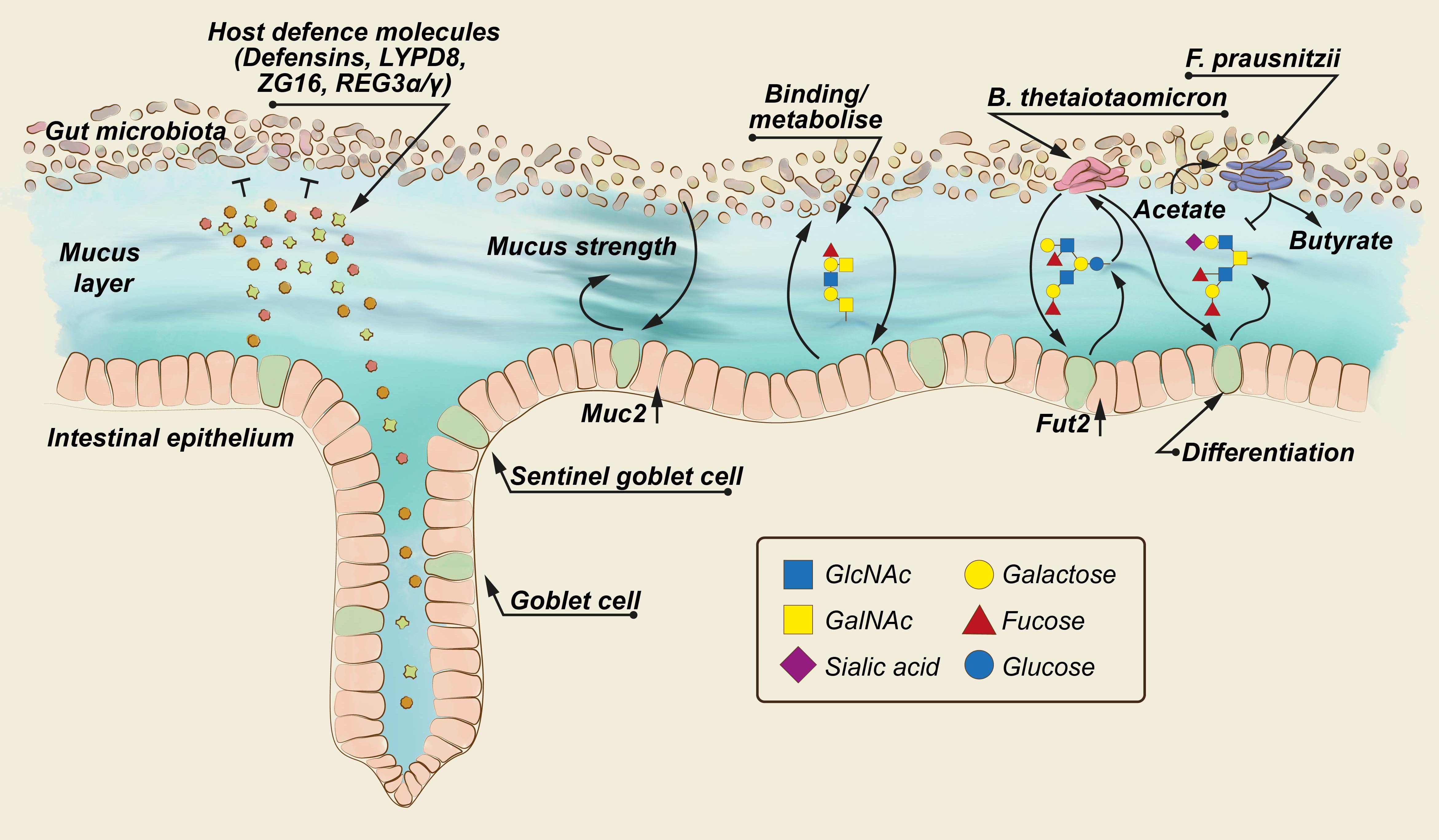
Over the course of evolution, the body has developed various mechanisms of border protection. First, a dense and sticky layer of mucus covers the intestinal surface and thereby physically hinders bacteria from invading into the body. In addition, mucus is filled with so called antimicrobial peptides, which are antibiotics produced by the body to kill bacteria that manage to penetrate into the mucus barrier.
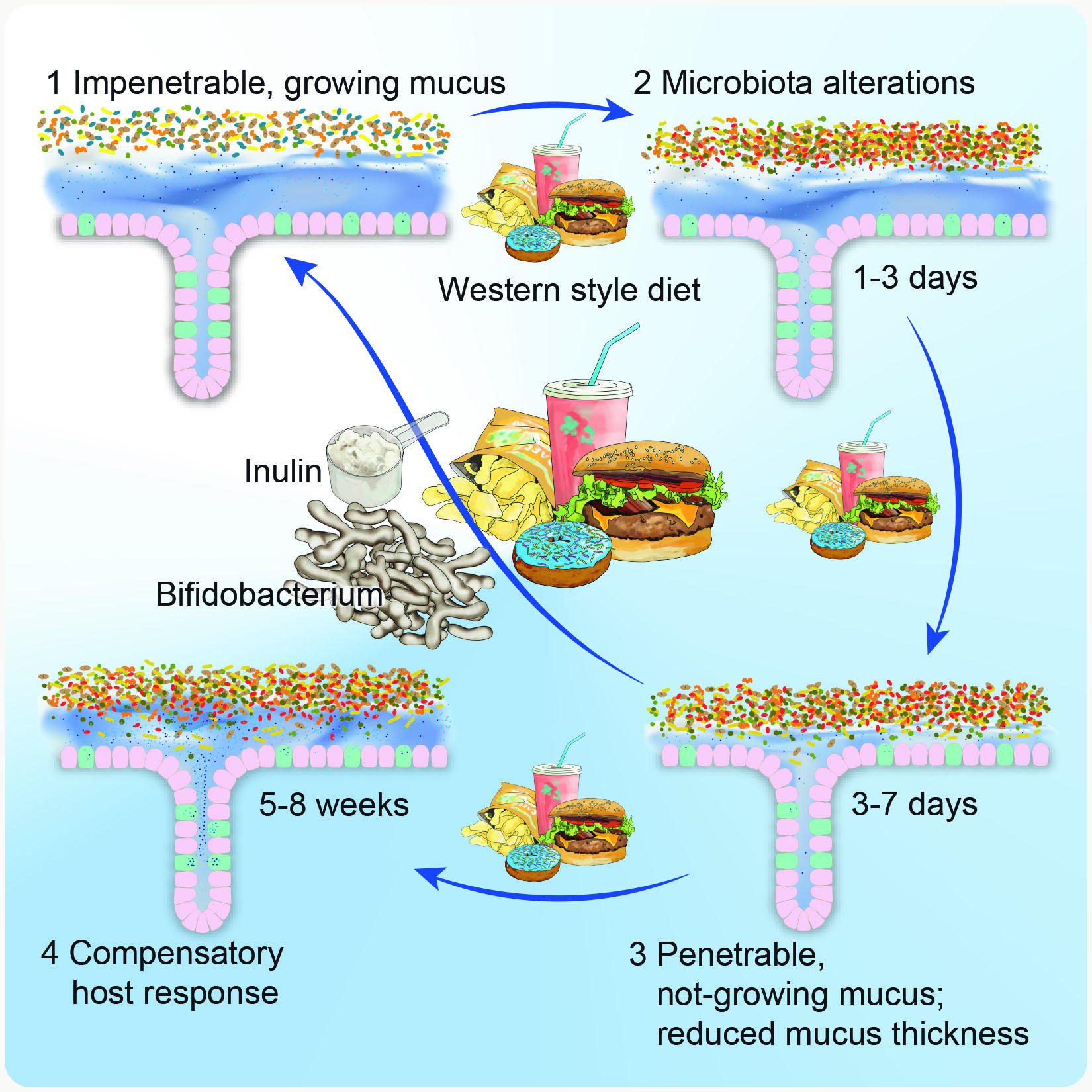
During previous studies in the labs of Fredrik Bäckhed and Gunnar Hansson we found that mice that were fed a “Western-style diet” – a diet similar to fast food that contains high amounts of simple sugars and fat but lacks dietary fibre – had a different composition of gut bacteria, which caused damages in the mucus shield (Schroeder BO et al., Cell Host & Microbe 2018). Consequently, bacteria could come closer to the epithelium, which increases the risk for infections and inflammation in the gut. Of note, we found that application of a probiotic Bifidobacterium or the prebiotic dietary fibre inulin, which is, for example, present in chicory and Jerusalem artichokes, was protective for intestinal barrier function.
In the MUCUBACTER lab we have during the recent years extended our research on human samples. By transplanting human gut bacteria, derived from population-based biobanks or diet interventions, we found that antibiotic intake leads to microbiota-mediated mucus defects (Krigul K, Feeney RH et al., Gut Microbes 2024) and that increasing dietary fiber intake in humans shapes their microbiota into a configuration that prevents mucus function (in mice) under Western-style diet feeding. (Holmberg SM, Feeney RH, Prasoodanan PKV et al., Nat. Communications 2024).
While we even identified the molecular mechanism of how the commensal bacterium Blautia coccoides modulates mucus function, many detailes of the diet-microbe-mucus interactions are yet to be determined – one major aim of our research!
Antimicrobial peptides are the second research focus in the lab. These host-produced peptide antibiotics keep bacteria at a distance from the epithelium, and are also thought to shape the composition of the gut microbiota. However, in a carefully controlled study we could not see a strong microbiota-modulating effect through AMPs, at least when compared to diet Puertolas-Balint F & Schroeder BO., Microbiol. Spectrum 2023). Yet, we identified several factors that affect AMP expression in the samll intestine, and could even identify a new AMP-inducing bacterium Puertolas-Balint F et al., Gut Microbes 2025).
Through a combination of in vitro and in vivostudies we continue to better undertsand the interaction beteen intestinal AMPs and the the gut bacteria.
With our research we want to contribute to a better understanding of how modern dietary habits affect intestinal defence mechanisms.
In the industrialized world, diseases such as inflammatory bowel disease and metabolic disease are on the rise and have been linked to the gut microbial community and intestinal barrier defects. As diet is one of the most important factors that shapes the community in the gut, we hope to clarify the involvement of specific food components in the complex regulation of gut barrier function. Besides contributing to improved dietary recommendations for the Western society, we hope to identify important bacteria that can be used as future probiotic supplements to improve barrier function in the gut. Moreover, we hope to find nutritional compounds that can enrich these positive bacteria to provide long-term health benefits for the Western gut.
